REGULATORY SERVICES

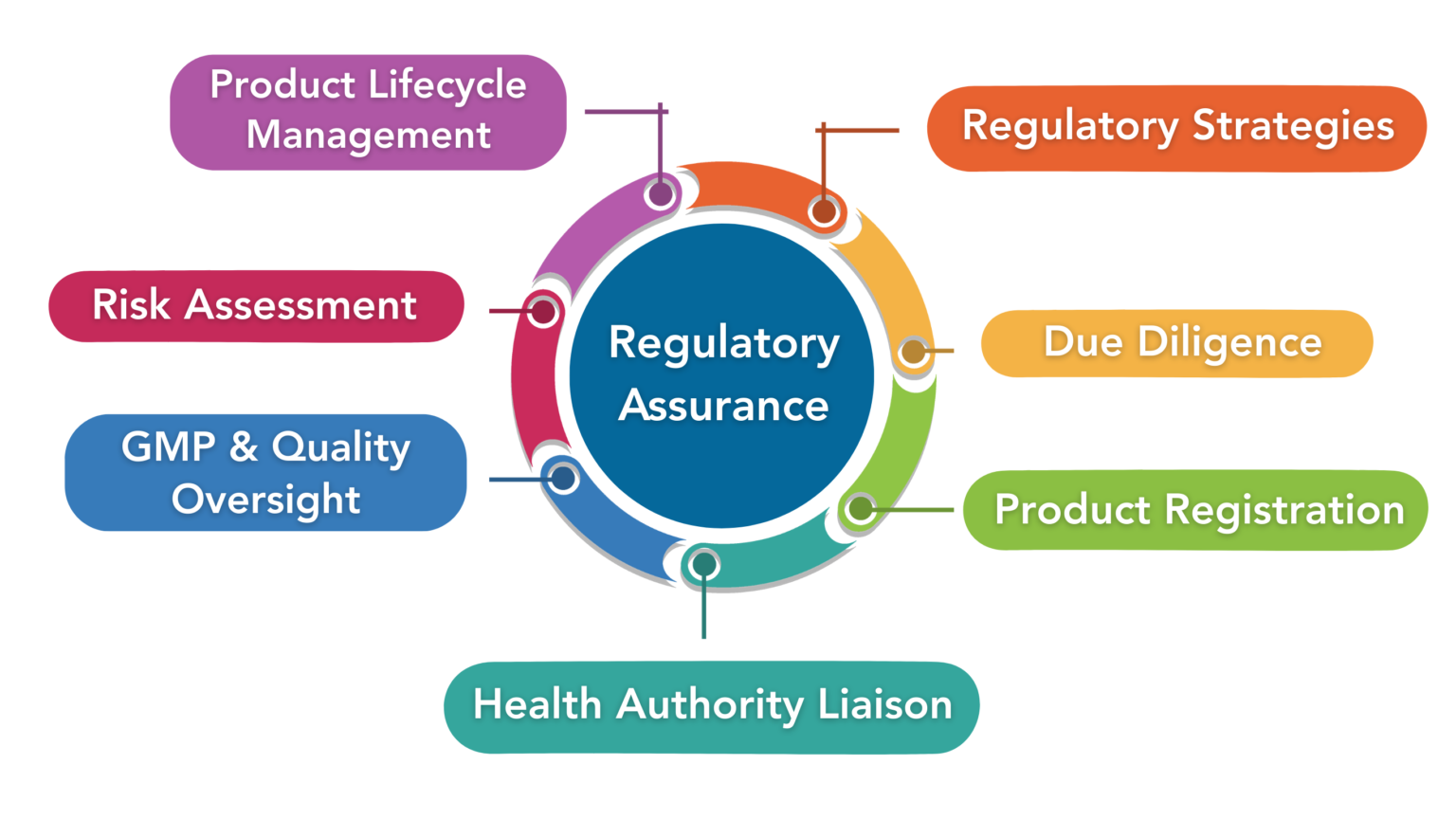
Propharmex specializes in end-to-end regulatory services, supporting companies in achieving Regulatory strategic pathways and timely approvals with full compliance for prescription drugs, natural health products, and medical devices.
We offer strategic, time-sensitive consultation on regulatory and GMP compliance, including complete management of compliance audits and assistance in securing Drug Establishment Licences (DEL) for importation and distribution when it comes to Health Canada. Through our strategic alliance with Canadian Healthcare Logistics (3PL) Propharmex delivers seamless, one-stop solutions for all your regulatory, compliance, and warehousing needs.
Regulatory Services
Propharmex specializes in end-to-end regulatory services, supporting companies in achieving Regulatory strategic pathways and timely approvals with full compliance for prescription drugs, natural health products, and medical devices.
We offer strategic, time-sensitive consultation on regulatory and GMP compliance, including complete management of compliance audits and assistance in securing Drug Establishment Licences (DEL) for importation and distribution when it comes to Health Canada. Through our strategic alliance with Canadian Healthcare Logistics (3PL) Propharmex delivers seamless, one-stop solutions for all your regulatory, compliance, and warehousing needs.
Regulatory Strategy & Market Access
CMC Authoring and Publishing
Quality System & Good Manufacturing Practices
Post Market Compliance Services
Clinical Services & Regulatory Support

Regulatory Strategy & Market Access
Concept to Commercialization
Regulatory complaint pathways
Minimize delays/Maximize success

CMC Authoring & Publishing
High quality CMC authoring
Maintain product lifecycle
Tailor made data presentation
Ensuring accuracy, compliance, efficiency

Quality System & Good Manufacturing Practices
End-to-end quality and GMP support
DEL/GMP audit readiness
3PL logistics

Post Market Compliance Services
Continued Market access
Post market compliance
Pharmaco Vigilance

Clinical Services & Regulatory Support
End-to-end clinical study support
Strategic scientific guidance
Robust documentation, Regulatory complaint
Partner with us to address your specific needs and achieve your strategic business goals
Your vision, our expertise. driving results that matter
