ANALYTICAL SERVICES
ANALYTICAL SERVICES

Method Development
Optimizing analytical methods to identify or quantify substances, often using techniques like HPLC, GC, and UV-Vis.

Method Verification & Validation
Ensures that an analytical method is suitable, reliable, and reproducible for its intended purpose for regulated markets like the FDA, EMA, and ICH. Compendial methods shall be verified for their suitability to
test the drug product.

Method Transfer
To ensure that the method performs equivalently in the receiving laboratory

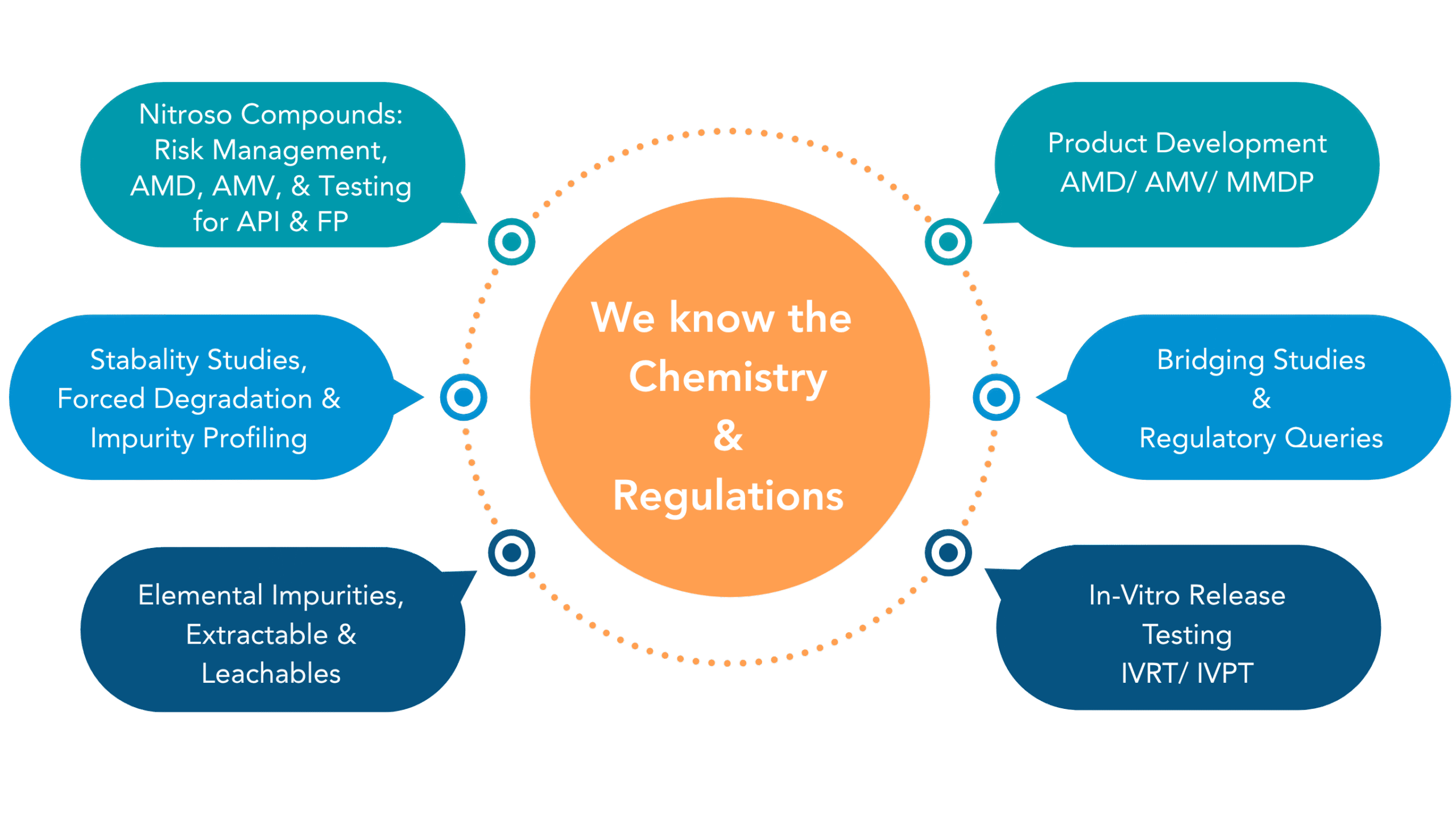
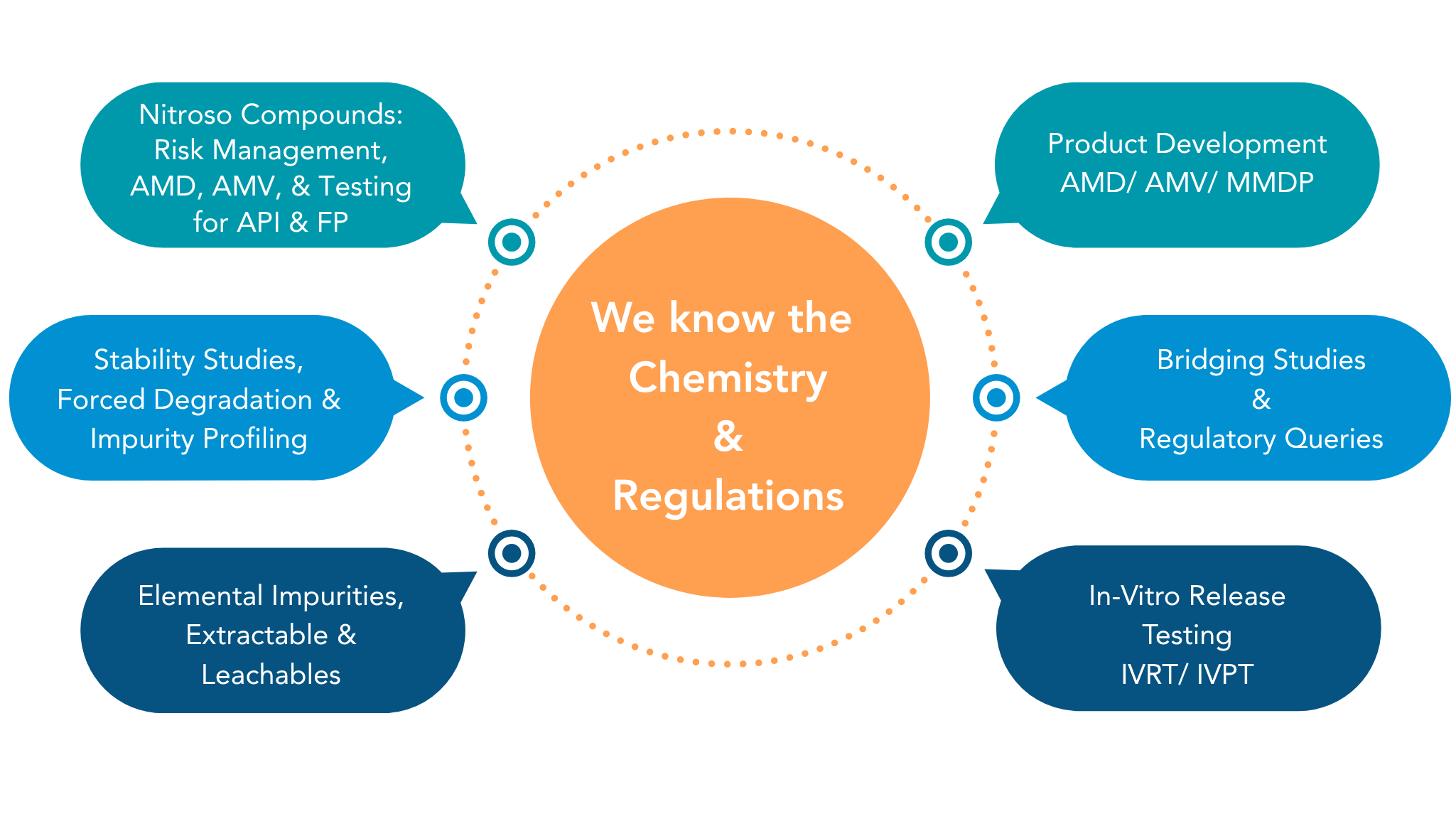
Impurity & Degradant Profiling
Any unwanted chemicals that may remain with the active pharmaceutical ingredient (API) during synthesis or API undergoing stress during manufacturing process of drug product & shelf life may lead to degradants.

Batch Analysis and QC Testing
Release Testing of Raw Materials (APIs, & Excipients) & Drug Products.

Stability Studies
To test the product retains its identity, potency, purity, safety,
and efficacy for intended duration at defined storage conditions and establish shelf life.
- Dissolution apparatus
- HPLC with PDA detector
- HPLC with UV detector
- UV- Visible Spectrophotometer
- Stability Chambers
- Semi-Micro Balance
- Micro Balance
- Rheometer
- Modular Microscope
- Vertical Franz diffusion Cells
- Viscometer
- Immersion cell
Explore Our Specialized Analytical Services
Discover data-driven insights tailored to your needs. Our specialized services turn complexity into clarity. click below to explore how we can empower your decisions.
Partner with us to address your specific needs and achieve your strategic business goals
Your vision, our expertise. driving results that matter
